Introduction
Juxtaglomerular cell tumors, also termed reninomas, are rare kidney tumors that usually present at a young age with symptoms of secondary hyperaldosteronism.1 Hypertension with or without hypokalemia in combination with a small renal mass should warrant the according diagnostic work-up. Reninomas are generally considered benign tumors, however, single cases of metastatic disease have been reported.2 Open or laparoscopic surgical removal is the treatment of choice and is considered curative in the overwhelming majority of cases.3 So far, only one report of reninoma resected with robotic-assisted surgery has been published.4 We present the case of a young female patient, with a 10-year long history of hypertension, who underwent robotic-assisted partial nephrectomy for a reninoma. In this paper, we describe the diagnostic evaluations, histological features and surgical removal of this rare tumor.
Case presentation
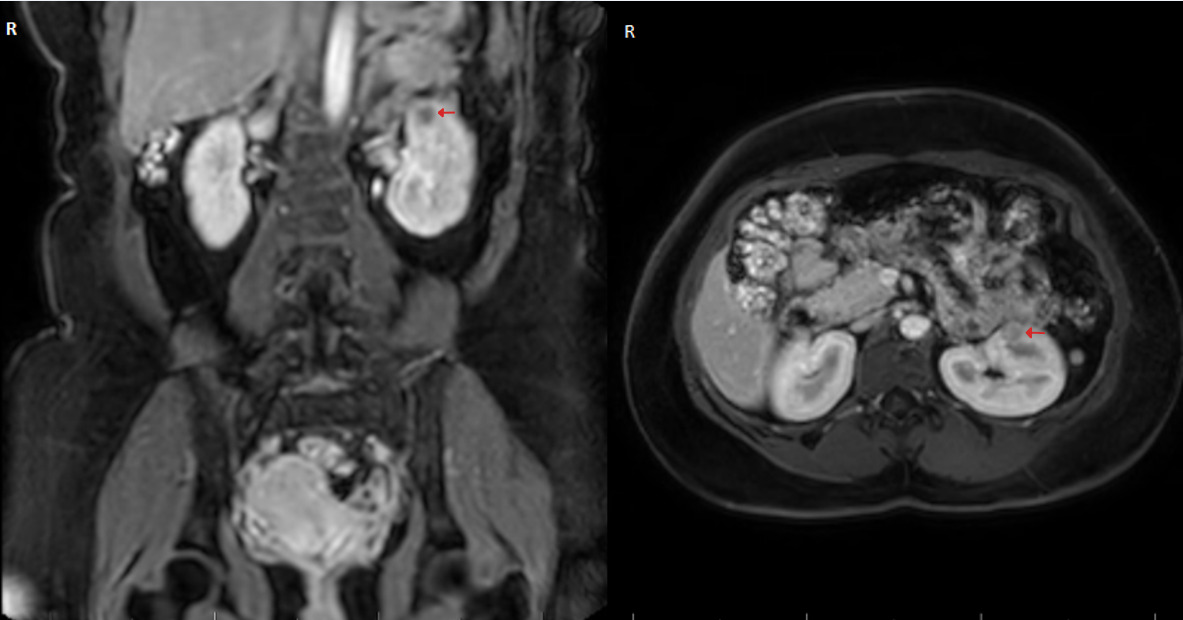
A 32-year-old woman was referred by her general practitioner to our urology department for the surgical removal of a small renal mass. Her medical history was remarkable for hypertension first diagnosed at 22 years of age, which was medically well controlled with amlodipine and enalapril. The patient reported that during the diagnostic workup of her hypertension, an elevated renin and aldosterone serum level had been noted, although the exact values could not be retrieved as the tests were performed in a different country. An external magnetic resonance imaging (MRI) in 2019 revealed a small renal mass of 8x9 mm in size in her left kidney, which was followed up with a second MRI in November 2022, revealing an increase in size to 16x13 mm and suspicion of malignancy (Figure 1). Hematuria or flank pain were denied, as was urinary discomfort or an increase in urinary frequency. Her medical history for previous abdominal surgeries or allergies was void. The physical exam revealed a soft abdomen without a palpable mass. The laboratory findings demonstrated adequate renal function with a serum creatinine of 57 µmol/l and a glomerular filtration rate (Cockroft-Gault) of 118 ml/min/1.73 m2. In the perioperative screening potassium levels were normal. According to the patient hypokalemia was also absent in previous blood tests.
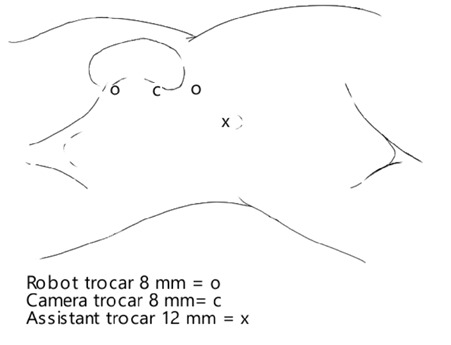
A robotic-assisted resection of the tumor in the left kidney (the daVinci Xi Robotic Surgical System® by Intuitive Surgical®) was performed on December 21, 2022. A Veress needle was used for the induction of pneumoperitoneum. Afterwards, trocar placement was performed as shown in Figure 2. The robot trocars are placed in a linear fashion on the lateral border of the musculus rectus abdominis. The exophytically-growing tumor is easily identified in situ. After adhesiolysis between the surrounding fatty tissue with the peritoneum and the caudal pancreas, the adipose tissue surrounding the tumor was carefully removed and sent to the pathology department for histological analysis, exposing the tumor. The resected tissue was extracted via the 14 mm trocar using an endobag. The margin delineating the tumor from the healthy tissue was clearly identified. The resection margin was sonographically determined and the tumor was resected with a 2 mm margin. Due to robotic assistance, iatrogenic ischemia of the kidney was not necessary. The total blood loss was minimal (90 ml) and the procedure was completed in 1 hour and 47 minutes.
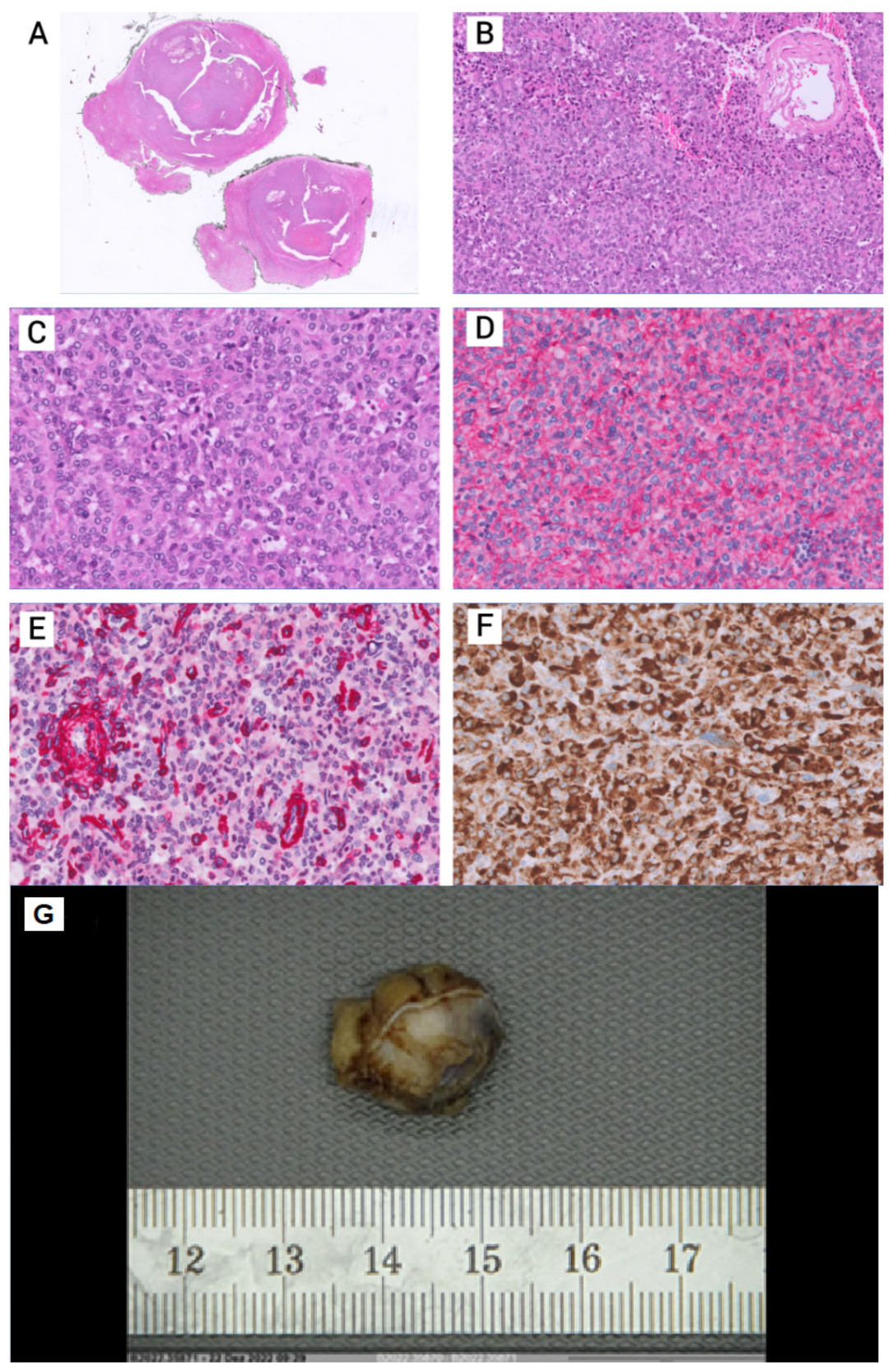
The histological specimen showed a juxtaglomerular tumor without signs of malignancy, with clear resection margins. The tumor was well-vascularized and composed of polygonal cells with eosinophilic cytoplasm. Necrosis and relevant cytological atypia were absent in the resected tissue (Figure 3 A-C and F). Lesional cells revealed a strong reactivity for the vascular marker CD34 (Figure 3 D). Immunohistochemistry demonstrated positivity for renin (Figure 3 F).
The patient’s convalescence was without complications, blood pressure returned promptly to normotensive values of (average 120/75 mmHg) without the need for continuing antihypertensive medication. The patient was discharged after 3 days.
Discussion and literature review
Juxtaglomerular cell tumors secreted an excessive amount of renin, which causes secondary hyperaldosteronism and consequently hypertension.5,6 Due to its rarity, the time it takes to reach a confirmed diagnosis is often extended.
While hypertension itself is one the most common chronic diseases, the prevalence of secondary hypertension is estimated at 5−15%.7 The primary culprits of secondary hypertension are obstructive sleep apnoea, renal artery stenosis, primary aldosteronism and thyroid disease.7 Screening all patients for secondary hypertension is not recommended.8 However, our case underscores the necessity of promptly ruling out secondary causes of hypertension in young patients, in accordance with international guidelines.8 Indeed, younger patients <40 years of age with grade II hypertension (e.g., with a blood pressure level of 160−179/100−109 mmHg) as well as the onset of hypertension in childhood should warrant the search for other identifiable causes.
Measuring plasma renin concentration and renin activity should be conducted for all patients with suspected hyperaldosteronism. In the past hypokalemia was described as hallmark feature of hyperaldosteronism; more recent findings showed decreased levels of serum potassium in only about 16% of cases. Our case highlights the fact that primary and secondary hyperaldosteronism often presents without hypokalemia.9 Radiological imaging can be employed to identify underlying conditions associated with hyperaldosteronism, such as adrenal hyperplasia, endocrine tumors, or renal artery stenosis.10
Reninoma is considered an extremely rare tumor entity, first described in 1967,11 with approximately 100 cases reported in current literature.5 As malignant tumors are far more common causes of small renal masses12 and imaging findings can be variable,13 a surgical approach suitable for a potentially malignant neoplasm is warranted. Currently, open or laparoscopic nephron-sparing, mainly partial nephrectomy, are the most common surgical procedures.14 Our urological department prefers a robotic-assisted approach for minimal invasive kidney surgery. The advantages of this procedure are a lower perioperative complication rate, lower risk of conversion to an open procedure, lower blood transfusion rates and shorter hospitalization.15
Due to the easy abdominal entry and exploration of the tumor, we decided against the use of four robot trocars, which is the standard setup for robotic-assisted kidney tumor enucleation and partial nephrectomy in our institute. The trocar count and configuration should be chosen, among many other aspects, based on the skill of the personnel involved, tumor size and the anatomical features of the patient. Possible cosmetical benefits are secondary to the patient’s safety.
Conclusion
The case is remarkable for the long history of hypertension in this young patient in the run-up to the finding of the renal mass. Robotic-assisted partial nephrectomy was feasible and safe in this patient suspecting a juxtaglomerular cell tumor of the kidney. Open or laparoscopic resection are the current standard of care. Due to the procedure’s advantages, a robotic-assisted approach may be considered as a favorable alternative in selected patients.
Acknowledgments
We thank Dr Tarun Mehra for editorial support.
Ethics approval and consent to participate
Not applicable.
Consent for publication
General written informed consent was obtained from the patient for the publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Availability of data and materials
All patient data that support this case report are included in anonymized form in the published article.
Conflicts of interest
All authors have declared that they do not have any conflict of interest and that they have no financial relationships with any organizations that might have an interest in the submitted work.
Funding
All authors have declared that no financial support was received from any organization for the submitted work.
Author contributions
J. Engesser wrote the manuscript. K. Mertz and J. Haslbauer performed the histological examination of the tumor. S. Subotic and P. Spoerri supervised the project. All authors contributed to and approved the final manuscript.