Background
Colorectal cancer (CRC) is the second most common solid cancer in women and the third most common in men, accounting for approximately 10% of new malignant tumors and cancer-related deaths globally.1 China estimated nearly 555,477 new CRC diagnoses and 286,162 deaths attributable to CRC in 2020.2 By 2030, more than 2.2 million new cases and nearly 1.1 million colon cancer-related deaths are expected worldwide.3,4 Patients with oncologic conditions are frequently among the most vulnerable patient populations because of their heightened risk for opportunistic and other infections or medical complications, with immune deficiency as an important factor to be considered. Active preparation of efficient and safe healthcare delivery for such patients is critical during the ongoing pandemic, beyond a simplistic assumption of chemotherapy regimen reduction or pausing to avoid excessive immunosuppression.5
In March 2020, the Ministry of Health in China issued coronavirus disease 2019 (COVID-19) pilot diagnosis and treatment guidelines, “COVID-19 diagnosis and treatment Program (7th edition)”.6 The guide provided information on COVID-19 pathology and updated information about clinical manifestations, diagnostic criteria, treatment methods and discharge criteria, compared with the 1st edition issued in February 2020. As a national-level guideline, it was adopted by all clinical institutions in China that provide routine and ongoing patient care, including oncology patient management with surgery and chemotherapy.
This article aims to share the experience and insights learned from a representative academic central top-tier medical center during the COVID-19 pandemic, including the hospital triage system for all the patients, the CRC treatment strategy and the application of telemedicine in follow-up care and clinical consultation. The concise experience which yielded a rapid and successful turnover of patients during the quarantine period and return to an even more efficient system thereafter may offer strategies for healthcare organizations to plan and deliver oncology patient care during emergencies, such as a pandemic.
Brief introduction of COVID-19
COVID-19 first appeared in December 2019 and was caused by a type of novel coronavirus, then known as a 2019-new coronavirus (2019-nCoV), with the disease named coronavirus disease-2019 (COVID-2019) by the World Health Organization (WHO).7 Later, the International Committee on Taxonomy of Viruses renamed 2019-nCoV to severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2).8 SARS-CoV-2 is primarily transmitted through droplets or direct contact among humans. After exposure, there is a 2–14-day incubation period with an average of 5 days.9 The basic reproduction number was estimated to range between 2.24–3.5810 and 2–6.47.11,12 During the incubation period, viral transmission to another person may occur.10
Fever is the primary manifestation in most cases but initial clinical symptoms may also include cough, dyspnea, myalgia, fatigue, cephalgia, diarrhea, sore throat, rhinitis and, in some patients, prominent anosmia and dysgeusia.13–16 Patients with underlying conditions, such as cardiovascular disease, hypertension, obesity and diabetes mellitus, are individuals at high risk for severe COVID-19.13,17 Patients with severe COVID-19 routinely develop dyspnea.14,15 Those who rapidly develop acute respiratory distress syndrome (ARDS) also clinically decline quickly, resulting in often fatal multi-organ failure.15 COVID-19 infection and mortality rates vary globally due to multifactorial reasons.
Methods
From December 2019 to February 2020, all patients who received medical care at Shanghai East Hospital, regardless of diagnosis, were required to strictly follow a hospital triage system established to mitigate the spread of COVID-19. For inpatient hospitalizations, all patients were required to have a negative polymerase chain reaction (PCR) test for SARS-CoV-2 before being admitted to the hospital. Nurses supervised and guided the triage system. For CRC patients, a combination of COVID-19 triage and general guidelines from the National Comprehensive Cancer Network (NCCN),18,19 European Society for Medical Oncology (ESMO)20 and Chinese Society of Clinical Oncology (CSCO)21,22 for CRC were used to develop a new management strategy. Since February 2020, all healthcare workers have undergone SARS-CoV-2 PCR testing twice a month. The number of inpatient hospitalizations and surgeries at Shanghai East Hospital and for the Department of Gastrointestinal and Colorectal Surgery during this period were collected. The triage system and management strategies at our institution were established as described below.
Triage system for patients during COVID-19
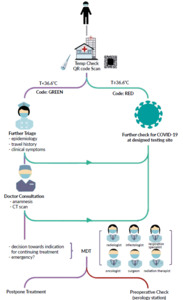
To mitigate COVID-19 transmission during routine medical care, a triage system was established by epidemiologists, respiratory experts and hospital managers (Figure 1) at Shanghai East Hospital located in Lujiazui, Shanghai, China, a large general hospital affiliated with Tongji University with more than 1,800 inpatient beds. Experts combined clinical experience and mandatory control measures from the government. This triage system was applied to all clinical care. At an individual level, a limited number of persons, no visitors and only one accompanying person were allowed per patient. Each incoming patient and their accompanying person were required to wear a mask and undergo screening at the external hospital gate. The screening included a temperature check and scan of their national electronic permit QR code.
The government designed the national electronic permit QR code, containing personal information like gender, age, address and health status. These data are associated with authoritative data from the local Ministry of Health, including whether there are any cases of COVID-19 that are new, suspected or confirmed. A suspected case is defined as an individual who had contact with a confirmed case within 14 days, has symptoms potentially consistent with COVID-19 or had a positive SARS-CoV-2 antibody test that has not been confirmed by nucleic acid detection. A confirmed case is defined as an individual with a positive SARS-CoV-2 PCR test.
Data are processed, considering if an individual has been in contact with confirmed or suspected COVID-19 cases, and then a QR code is generated with color codes as a “risk hint”. Red represents a positive risk: the individual has suspected/confirmed COVID-19 or had contact with a suspected/confirmed case within 14 days. Green represents no risk: there are no symptoms potentially consistent with COVID-19 and no contact with a suspected/confirmed case within 14 days. At the screening for hospital entry, only the green QR code is permitted for entry or admission. Those with a red QR code will be advised to be isolated; if a SARS-CoV-2 PCR test is positive, the patient will be escorted to a specific infectious disease hospital specializing in treating COVID-19 patients.
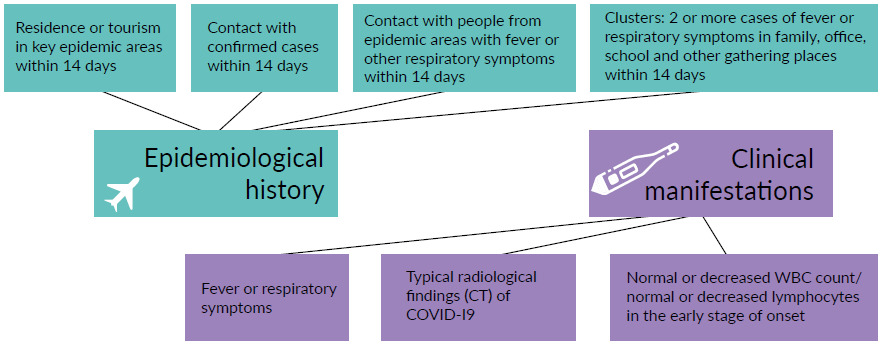
Upon entering the hospital, each patient underwent a comprehensive anamnestic interview with a practicing nurse on a designated floor located apart from other departments. According to the official guiding classifications, in-depth epidemiological history and clinical symptoms were documented to evaluate for suspected COVID-19 cases (Figure 2).23 If patients had unremarkable interviews with the nurse, they proceeded with their physician consultation appointment. However, patients with any remarkable items on their anamnestic interview (i.e., one of the items of epidemiological history, any two clinical manifestations or no epidemiological history but three clinical manifestations) were triaged for further etiological testing, including reverse transcription polymerase chain reaction (RT-PCR) detection of SARS-CoV-2, viral gene sequencing and antibody detection (Figure 1). For suspected cases presented during an emergency, the patient’s condition was carefully evaluated and immediate surgical treatment was arranged in designated centers.
Throughout this triage system, clinical staff used secondary personal protective equipment (PPE) during patient care delivery (Table 1). Such secondary PPE offered greater protection compared with primary PPE. These precautions were intended to effectively reduce the probability of body fluids coming into contact with the eyes and respiratory mucosa of clinical staff during examination.
Management strategies for patients with CRC in our institution during the COVID-19 pandemic
a) Evaluation
Prior to COVID-19, all CRC patients requiring surgery underwent a routine preoperative examination to evaluate their cardiopulmonary function and general physical condition. During the COVID-19 pandemic, examinations such as colonoscopies, biopsies and radiological imaging (abdominal and pelvic computed tomography [CT]/magnetic resonance imaging [MRI], positron emission tomography [PET]-CT) were limited as diagnostic/evaluation tools. For example, they were no longer used for CRC screening. If COVID-19 was neither suspected nor confirmed, the preoperative evaluation (including circulatory system function examination, respiratory system function examination, blood coagulation function test, etc.) remained unchanged. However, if COVID-19 was suspected, preoperative preparation was modified to include also a pulmonary spiral CT examination and SARS-CoV-2 test (nucleic acid and antibody detection). Invasive examinations like digital rectal examinations, colonoscopies and biopsies were carried out in a specific isolation room by clinical staff using tertiary PPE (Table 1) and all the examinations were carried out under the guidance of an infectious disease specialist. Such supplemental protective measures were adopted because of previously published data about the Middle East respiratory syndrome (MERS) that the virus can be transmitted by the fecal-oral route.24 SARS-CoV-2 nucleic acid has also been detected in the feces of confirmed COVID-19 cases.25,26
Multidisciplinary therapy meetings (MDT), already an essential component of routine oncologic patient care, were maintained and even strengthened during COVID-19. MDT consist of discussing the risks and benefits of various therapeutic options for an individual patient, drawing from the expertise of a variety of healthcare professionals, including, for example, department representatives from multiple specialties and disciplines such as general surgery, oncology, radiology and radiotherapy. During the COVID-19 pandemic, MDT also involved specialists in respiratory medicine, epidemiology and infectious disease. They provided expertise on patient risks in case of SARS-CoV-2 infection (e.g., lung reserve and preexisting organ damage).
b) Treatment and management
The Department of Gastrointestinal and Colorectal Surgery at Shanghai East Hospital had previously established an evidence-based treatment strategy for patients with CRC prior to the COVID-19 pandemic. Because the pandemic introduced additional potential risks for patients undergoing treatment, modified treatment strategies were considered in addition to standard care. Here, an overview is provided to highlight these treatment modifications for patients with CRC during COVID-19.
1. Neoadjuvant therapy
In the background of COVID-19, it was important to consider applying neoadjuvant chemotherapy (NAC) to CRC patients during the pandemic and not withhold this crucial therapeutic step, despite potentially increasing the infection risk. At present, the preoperative treatment for colon cancer is primarily NAC which can eradicate occult micro-metastatic disease at an earlier time, improve tolerability and dose intensity to the chemotherapy drug, and assess the response to chemotherapy to guide adjuvant considerations. Preoperative chemical tumor reduction may lower the frequency of tumor cell shedding during surgery and increase R0 resection rates. NAC can also improve postoperative recovery by minimizing the extent of surgery. Previous studies have shown that NAC is safe and effective for some cT4 patients and can improve the survival rate of patients.27–29 The NCCN guidelines also emphasize NAC with folinic acid, fluorouracil and oxaliplatin (FOLFOX) or capecitabine and oxaliplatin (CapeOX) as a potential treatment for resectable cT4b colon cancer.19,30 Currently, clinical trials have proved that both FOLFOX regimen and CapeOX regimen have good efficacy in the treatment of locally advanced colon cancer.29,31 An American National Cancer Database (NCDB) analysis further suggested that neoadjuvant radiotherapy (NAR) could be considered as an alternative in locally advanced cT4 colon cancer, but only on a case-by-case basis.32 Patients with no further major comorbidities, a Karnofsky score of 0 or 1 and younger than 65 years were preferred to receive the therapy in an outpatient setting.
The application of radiotherapy in rectal cancer is more beneficial than in colon cancer. Several existing studies have shown that preoperative neoadjuvant chemoradiotherapy (NCR) for cT3-4 rectal cancer has significant benefits in reducing the local recurrence rate compared with postoperative chemoradiotherapy. It also has significant benefits in improving local control anus retention rate and is associated with reduced toxicity compared with postoperative chemoradiotherapy, despite no significant difference in overall survival.33–38 Additionally, preoperative neoadjuvant chemoradiotherapy can reduce tumor size, increase the tumor resection rate and anus retention rate with very mild side effect.39 Both NCCN and ESMO guidelines regard preoperative simultaneous radiotherapy and chemotherapy plus total mesorectal excision (TME) surgery plus adjuvant chemotherapy as the preferred treatment strategy for locally advanced cT3-4 rectal cancer.18,19,40,41 Therefore, in our department, we recommend that neoadjuvant therapy (NAT) should be applied in patients with locally advanced colon cancer in stage cT4 and rectal cancer in stage cT3-4 without significant risk of bleeding, obstruction and perforation.
During the COVID-19 pandemic, the CapeOX regimen was considered to be more suitable because of its convenience, as the patients spend less overall time in the healthcare institutional setting compared with the FOLFOX regimen, resulting in potentially lower exposure risk to COVID-19.42 We followed the current NCCN guidelines, in which it is permitted that advanced rectal cancer is treated with radiotherapy, combined with oral capecitabine NAT.18 Whether surgical treatment should follow is evaluated 8 weeks later. For patients with primary liver metastasis of CRC, NAC can be considered first and after evaluation of the patient’s condition, resection of the primary focus combined with the metastatic focus can be performed. If the metastases are unresectable and the patients with metastatic CRC who are HER2-amplified, or BRAF V600E mutation-positive, NAC combined with targeted therapy can be considered.40
2. Endoscopic therapy
Endoscopic treatment is the first choice for early CRC patients (cT1N0M0) with a maximum diameter of the tumor of <3 cm. After endoscopic resection, a pathological examination must be performed and the necessity for additional surgical treatment should be evaluated according to the pathological result.
For patients with CRC complicated with obstruction, an emergency surgery rarely achieves an R0 resection. Moreover, intestinal wall edema and poor intestinal preparation increase the difficulty of operation, so endoscopic stent implantation was considered. There is no significant difference in the 3-year disease-free survival rate and overall survival rate between endoscopic stent implantation and emergency surgery, even though the former has a higher recurrence rate.43 It has been reported that emergency colonic stent implantation has a shorter operation time, hospital stay and lower infection rate than emergency operation.44 As a bridge to surgery, stent implantation achieves the effect of decompression for emergency colon cancer patients with obstruction, thus shortening the length of hospital stay, improving anastomosis rate, avoiding neostomy, reducing short-term complications and improving long-term survival.45,46 Colonic stent implantation followed by surgery or NAC has been proven to be safe and effective.47,48 Preoperative comorbidity is associated with poor prognosis in postoperative cancer patients, and postoperative morbidity and mortality are higher among those undergoing emergency surgery.49 Considering the impact of COVID-19 on the function of lung, heart, liver, kidney and gastrointestinal,50–52 colonic stent implantation is a relatively safe and effective choice for these emergency patients, especially for confirmed and suspected cases of COVID-19.
3. Surgical treatment
Stage cT2-3 colon cancer and stage cT2 rectal cancer are indications for radical surgery. If COVID-19 is excluded, then the operation could be carried out in accordance with the CSCO, NCCN or ESMO guidelines. A recent multicenter observational study reported that infection with COVID-19 in the perioperative period was associated with significant mortality.53 Considering the pressure on high-dependency beds, many elective CRC surgeries have been delayed or even canceled. A study reported that nearly 40% of CRC cases would be postponed during the initial 12 weeks of the COVID-19 pandemic.54 However, a review suggested that elective surgery for CRC patients is not postponed longer than 4 weeks, as available evidence indicates that extended delays from diagnosis are associated with poorer outcomes.55 Our MDT experts also recommend minimizing complex surgeries, to shorten the length of stay and reduce possible complications (fever, anastomotic leakage, abdominal infection, etc.).
Emergency surgery should be carried out in time for CRC patients accompanied by bleeding, perforation or obstruction. The operation is performed in a negative pressure room which offers optimal protection to the surgeon.56 Repeatedly emphasized, the surgeons and anesthesiologists must be in tertiary PPE. For patients with ultra-low rectal cancer, ileostomy is an effective measure to reduce the possibility of postoperative anastomotic leakage that can lead to pelvic infection and fever.
4. Postoperative management
Patients with a negative test for COVID-19 during preoperative evaluation received usual postoperative management, including short-term observation in the postoperative ward, nutritional support and vital sign monitoring. During the COVID-19 pandemic, in addition to usual management, postoperative monitoring included more frequent body temperature checks and respiratory symptom monitoring. If a temperature greater than 37.3 ℃ occurred, clinical staff would check the patient’s respiratory symptoms while wearing secondary PPE and recheck the patient’s blood indicators, including blood cell analysis, inflammation indicators, urinalysis, chest x-ray and arterial oxygen saturation. If physical examination and these tests did not rule out pneumonia or other typical postoperative etiologies of fever, chest CT examination was pursued. Furthermore, if fever and respiratory symptoms are present, protocols for suspected COVID-19 cases were applied. These include a single isolation room, frequent body temperature monitoring and oxygen inhalation. Once the SARS-CoV-2 nucleic acid test is confirmed (within several hours), the patient is then transferred to another specially designed infectious hospital designed for collecting all the COVID-19 patients and meanwhile, postoperative management is carried out there. Visitors were not permitted during the postoperative period during the COVID-19 pandemic. As for postoperative adjuvant therapy for CRC patients, we referred to the NCCN guidelines and CSCO guidelines.21,22 For patients with stage II−III CRC cancer, postoperative chemotherapy is recommended, and CapeOX regimen is recommended because of its convenience.
5. Application of telemedicine in Shanghai East Hospital during the COVID-19 pandemic
The COVID-19 pandemic stressed traditional medical systems. Consequently, remote work for healthcare professionals became essential to the delivery of continuing care. For example, nutrition education and stoma management for almost all CRC patients after operation were converted from in-person to online or teleconsultation visits. Also, physicians at Shanghai East Hospital increased the time for teleconsultations for patient care. For example, oncologists doubled their time spent on teleconsultations to 4 hours per week (previously 2 hours/week) while gastrointestinal and colorectal surgeons increased teleconsultation time to 5 hours/week (previously 2 hours/week).
Results
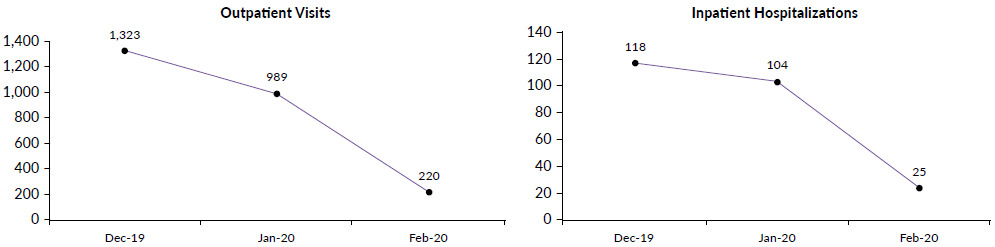
In February 2020, zero cases of COVID-19 have been identified among all inpatients and healthcare workers in Shanghai East Hospital (Table 2). Compared with the situation before the outbreak, the number of outpatients, inpatients and surgeries decreased considerably in Shanghai East Hospital, Tongji University School of Medicine (Figure 3) and at the Department of Gastrointestinal & Colorectal Surgery (Figure 4). Similar changes were also seen in the Department of Oncology (Figure 5).
Discussion
With the rapid spread of COVID-19, routine healthcare delivery services must have adapted to ensure high-quality care, safety and efficiency. The pandemic introduced numerous healthcare service delivery barriers, abruptly reducing the number of outpatient and emergency patients in the hospital and limiting inpatient surgeries, suggesting that a considerable number of patients did not get medical assistance in time. Patients with oncologic conditions like CRC faced unique challenges: they needed continuous medical care like chemoradiotherapy, however, they are also more susceptible to COVID-19 due to immunosuppression.57,58 Achieving a balance between the benefits and risks of delaying treatments for these patients during the pandemic is a major challenge for oncologists and surgeons.
The present case study suggests that such healthcare delivery modifications can help to resume oncologic care effectively during the COVID-19 pandemic. COVID-19 triage protocols, treatment modifications for patients with CRC who are at high risk for COVID-19-related complications and the use of telemedicine to continue providing care were key strategies in this case study for continuing care. As telemedicine has the advantage of reducing medical costs and increasing the availability of medical resources,59 it is widely used for patients who need continuous treatment, such as diabetes and oncologic diseases.59–62 It is believed that during COVID-19, the role of telemedicine will become more and more prominent. Such strategies during the pandemic permitted the continuation of CRC treatment, including neoadjuvant radiotherapy, neoadjuvant therapy, and endoscopic and surgical treatment.
Limitations of this report include generalizability, given that the case study provides the experience and data from only one region or hospital. Additionally, unique hospital and health system factors may reduce reproducibility. Adaptation and adoption of similar triage measures in other hospital systems may offer additional support to the effectiveness of the approach of Shanghai East Hospital. Further longitudinal evaluation of clinical and health system outcomes due to healthcare delivery modifications during the pandemic would be valuable.
Conclusions
The COVID-19 pandemic disrupted routine hospital operations and healthcare service delivery. The comprehensive triage and management strategy implemented at our hospital suggests that transmission control is possible, as zero new cases of COVID-19 were identified during the study period. Furthermore, applying management strategies based on clinical practice guidelines for patients with CRC also appears to have been beneficial, as zero COVID-19 cases were identified in this patient population. Nevertheless, managing CRC patients during the pandemic remains challenging. However, it seems more manageable with multidisciplinary, protocolized approaches integrating new technologies like telemedicine into healthcare delivery.
Ethics approval and consent to participate
The study was conducted according to the guidelines of the Declaration of China and approved by the Institutional Review Board of Shanghai East Hospital.
Consent for publication
Not applicable.
Availability of data and materials
The datasets generated and analyzed during the current study are not publicly available due these are the internal data information of Shanghai East Hospital but are available from the corresponding author upon reasonable request.
Competing interests
The authors declare no conflict of interest.
Funding
The research is funded by the National Natural Science Foundation of China (Grant No 82160515).
Author’s Contributions
Data analysis, writing and study design: Pengcheng Xiang; Data collection and investigation: Zitian Xiao, Xiuhao Zhao, Zhijin Zhang, Shaohua Qu; Review and editing: Tiffany I. Leung, Ewelina Biskup, Naomi I. Maria; Funding acquisition and study design: Junyi Han. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
Thanks for the help to the Department of Gastrointestinal and Colorectal surgery of Shanghai East Hospital, China.

_number_of_outpatient_visits_and_inpatient_hospitalizations_and_b)_number_of_surgeries_(.png)
_number_of_outpatient_visits_and_inpatient_hospitalizations_and_4b)_number_of_colorectal.png)


_number_of_outpatient_visits_and_inpatient_hospitalizations_and_b)_number_of_surgeries_(.png)
_number_of_outpatient_visits_and_inpatient_hospitalizations_and_4b)_number_of_colorectal.png)