Introduction
Modern antitumor immunotherapy represents one of the most actively developing fields of adoptive cell therapy. This approach involves the genetic modification of immune cells, such as T cells, natural killer (NK) cells and natural killer T (NKT) cells, to express chimeric antigen receptors (CARs) on their surface.1–4 CARs are synthetic constructs that, upon transfection into T lymphocytes, trigger a cascade of reactions and increased proliferative potential of CAR T lymphocytes, enabling them to specifically recognize and destroy tumor cells independently of the major histocompatibility complex (MHC) system. First-generation CARs consisted of a ligand-binding domain, typically derived from a monoclonal antibody, fused to the cytoplasmic signaling domain of CD3ζ.1,3 The incorporation of costimulatory signaling domains, such as CD28 or 4-1BB, has significantly enhanced the cytotoxic activity of CAR T cells, leading to the development and widespread use of second-generation CARs in current clinical protocols. Third-generation CARs, which combine multiple costimulatory domains, are also under investigation; however, their superiority over second-generation constructs remains to be established. The engineered nature of the CAR platform provides exceptional flexibility, permitting modifications to precisely tune CAR activity, persistence and cellular behavior.
Extensive research and clinical trials of CAR T cells are currently underway globally, with over 1,500 trials documented. Commercially available CD19-directed CAR T-cell therapies, such as axicabtagene ciloleucel, tisagenlecleucel and lisocabtagene maraleucel, have demonstrated high efficacy and durable remissions in patients with relapsed or refractory (R/R) B-cell acute lymphoblastic leukemia (ALL), chronic lymphocytic leukemia (CLL), non-Hodgkin lymphoma (NHL) and other B-cell malignancies.5–13 These CAR T-cell therapies have significantly improved clinical outcomes in patients refractory to standard treatments and are now part of routine clinical practice in many centers. However, their use remains limited by high acquisition and manufacturing costs, centralized production and restricted availability, particularly in countries with constrained healthcare resources.14 This necessitates the development of new CAR T-cell products and manufacturing strategies that can broaden access while maintaining its high quality and clinical effectiveness.
High costs and technological complexity remain major challenges for CAR T-cell manufacturing. The key stages of this complex, high-tech process include the production of lentiviral vectors, CAR T-cell culture, patient preparation and subsequent clinical administration with mandatory inpatient monitoring. Closed systems are being actively implemented for the production of genetically modified lymphocytes directly at the point of care, ensuring a high degree of sterility and process automation while reducing the time required to obtain the therapeutic product. Among these, the automated platforms Wukong (SinoBiocan, China) and KunLun (XinBio, China) stand out for their advanced technologies, reliability and scalability. Both systems provide robust platforms for cell therapy manufacturing and research, supporting all main stages of CAR T-cell production, including T-cell isolation, activation, lentiviral transduction, expansion and final processing (concentration and washing) within sterile closed circuits. By limiting open handling, this approach reduces the risk of microbiological contamination, minimizes operator involvement and accelerates the production process, enabling the standardization, optimization and scaling of cell processing operations. Local point-of-care production within a closed system also permits the use of leukapheresis samples without the need for complex logistics and cryopreservation, thereby preserving cell viability and potentially improving their functional performance.8 An important aspect in the development of CAR T-cell products is the assessment of immunophenotypes, antitumor activity and transduction efficiency in vitro.
This study presents the functional characterization of anti-CD19 targeting CAR T cells produced using the Wukong and KunLun systems focusing on immunophenotyping, evaluation of transduction efficiency and in vitro cytotoxicity.
Materials and Methods
Operational characteristics of the Wukong and KunLun cell processing systems
The Wukong cell processing system manufactured by Sino-Biocan (Figure 1A) is a new-generation, fully automated, self-contained, closed cell-processing platform that integrates multiple unit operations within a single instrument. The system can be configured to perform cell isolation, cell sorting, gene delivery steps such as transfection and transduction, cell culture, as well as concentration, purification and final filling of the cell product.
The SophCyte® KunLun fully automated cell preparation machine manufactured by Suzhou XinBio Co., Ltd. (Figure 1B) is a modular platform that can be used to obtain highly purified immune or stem cells from various types of starting material such as blood, bone marrow or other cell samples. By integrating different functional modules, the system generates a final cell product for use in cell therapy and other downstream applications.
Although both devices perform similar functions and processing steps, they differ in several key operational and performance characteristics (summarized in Table 1).
Automated manufacturing of clinical-grade CD19 CAR T cells using Wukong and KunLun cell processing systems
In this study, a leukapheresis sample obtained from a patient with B-cell lymphoma was used as the starting material. The sample was split into two aliquots under aseptic conditions for parallel processing on the Wukong and SophCyte® KunLun platforms. The biomaterial was processed within 24 hours of donation.
On day 1 of the production cycle, each leukapheresis aliquot containing the target number of T cells was diluted in platform-specific buffer (Sino-Biocan PBS/EDTA for Wukong; Suzhou XinBio Co. Ltd. buffer for KunLun), each supplemented with 0.5% human serum albumin (HSA; Takeda, Austria). The diluted samples were sequentially transferred to a set of sterile tubes supplied with the CD4 and CD8 microbead-based T-cell selection kits. For each platform, 1 × 108 isolated cells were activated in 70 ml T-cell culture medium containing 200 IU/ml recombinant human IL-7 and IL-15 and a CD3/CD28 microbead-based activation reagent. All platform-specific reagents, including T-cell selection kits, culture media and activation reagents, were obtained from T&L Biotechnology (China) for the Wukong system and from Suzhou XinBio Co. Ltd. (China) for the KunLun system. Cell activation was performed for 48 hours at 37 °C in 5% CO₂.
On day 3, activated T lymphocytes were transduced with a third-generation lentiviral vector (SuZhou XinBio Co. Ltd., China) at a multiplicity of infection (MOI) of 1 and co-incubated with the vector for 48 hours. Cells were then cultured for two additional days. On day 6, cells were washed and cultured on a shaker. On day 10 of the production cycle, cells were harvested, analyzed and cryopreserved in HSA supplemented with DMSO at -196 °C (Figure 2).
CD19 CAR expression during manufacture
T-lymphocyte purity and the distribution of T helper (CD4) and T killer (CD8) subsets were determined by flow cytometry using monoclonal antibodies against CD45, CD3, CD4 and CD8 (all from BD Biosciences, USA). T cells were identified using the following gating strategy: all cells → viable cells (7-AAD⁻) → CD45⁺/CD3⁺. CD19 CAR expression was detected using the CAR-detect-CD19 reagent (Artbiolent, Russia). Samples were analyzed using a BD FACSAria™ III flow cytometer (BD Biosciences, USA), with at least 0.3 × 106 cells recorded for each sample.
In vitro CAR T-cell cytotoxicity assay
Cytotoxicity was assessed using a fluorescence-based assay with the human Burkitt’s lymphoma-derived cell line Raji-mKate2 as a target. This CD19-positive cell line stably expresses nuclear-localized red fluorescent protein mKate2, which enables fluorescence-based quantitative monitoring of target cell viability and clear discrimination from non-fluorescent or differentially labeled effector cells in coculture. Raji-mKate2 cells can therefore be used to assess both antigen-specific CAR T-cell cytotoxic activity and non-specific cytotoxic activity using unmodified T cells as a control.
To assess the functional activity of CAR T lymphocytes and effector and target cells were co-cultured at CAR T:target ratios of 1:1, 5:1 and 10:1 for 24 hours. Cytotoxicity was quantified by fluorescence imaging using an Image ExFluorer system (LCI, Namyangju-si, Gyeonggi-do, Korea).
Results
CAR T-cell manufacturing output: Viability and yield
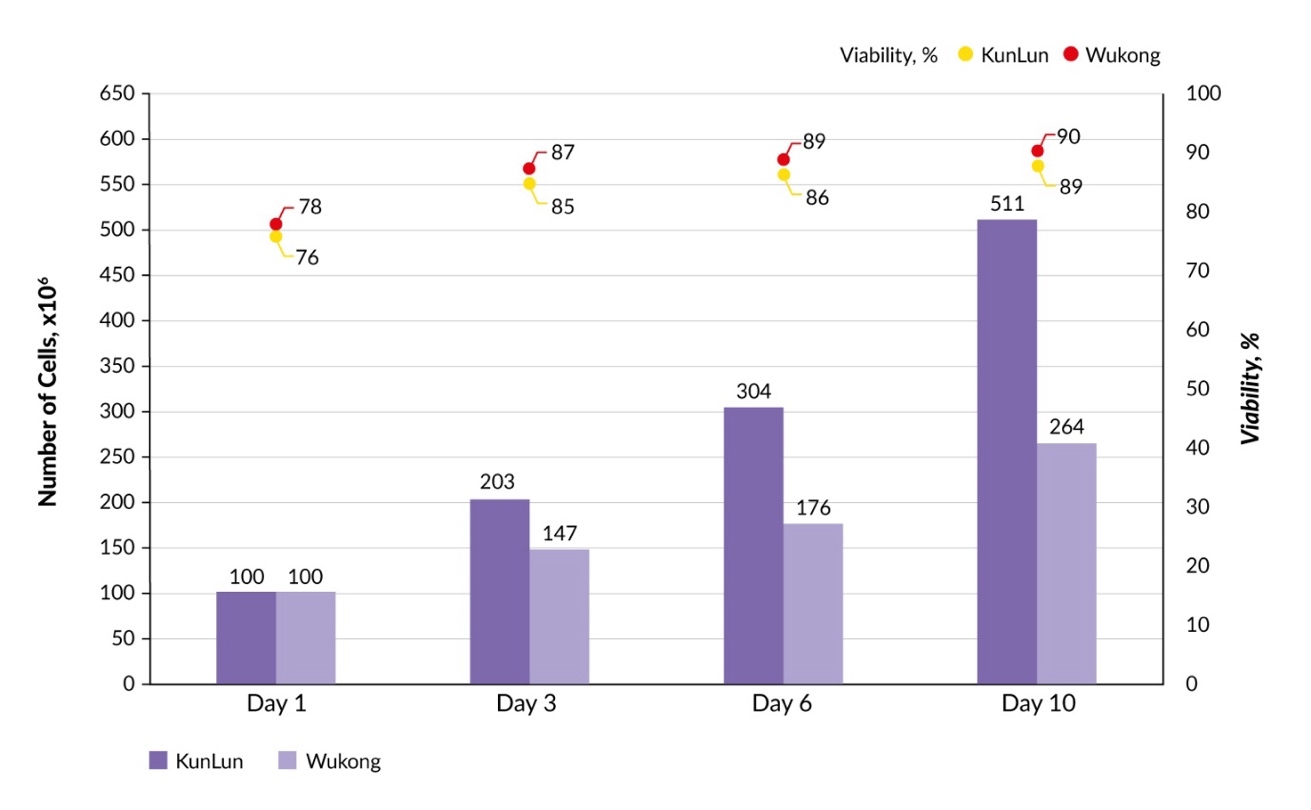
To compare CAR T-cell manufacturing on the Wukong and KunLun platforms, two CD19 CAR T-cell products were generated in parallel from the same leukapheresis starting material. Following T-cell selection and stimulation, cells were transduced with a lentiviral vector encoding a CD19-directed CAR. The production processes on both instruments were completed with minimal manual handling compared with conventional workflows. A total of 1 × 108 target cells were selected on each platform during the activation and transduction steps.
Cell count and viability were assessed during and at the end of the production cycle. Cell viability remained consistently high throughout the culture period, exceeding 90% on both platforms (Figure 3). Both systems supported substantial cell population growth; however, cell yield in the final product differed between the platforms. On day 10, the KunLun run yielded a total of 5.11 × 108 cells, compared with 2.64 × 108 cells obtained using the Wukong system. Thus, while both platforms generated viable, high-quality CAR T-cell products, the KunLun platform demonstrated greater efficiency in terms of cell mass accumulation under the tested conditions.
Transduction efficiency and immunophenotype characterization of generated CD19 CAR T cells
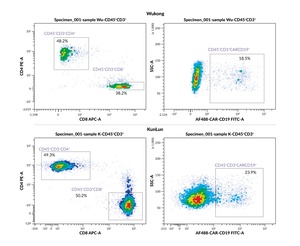
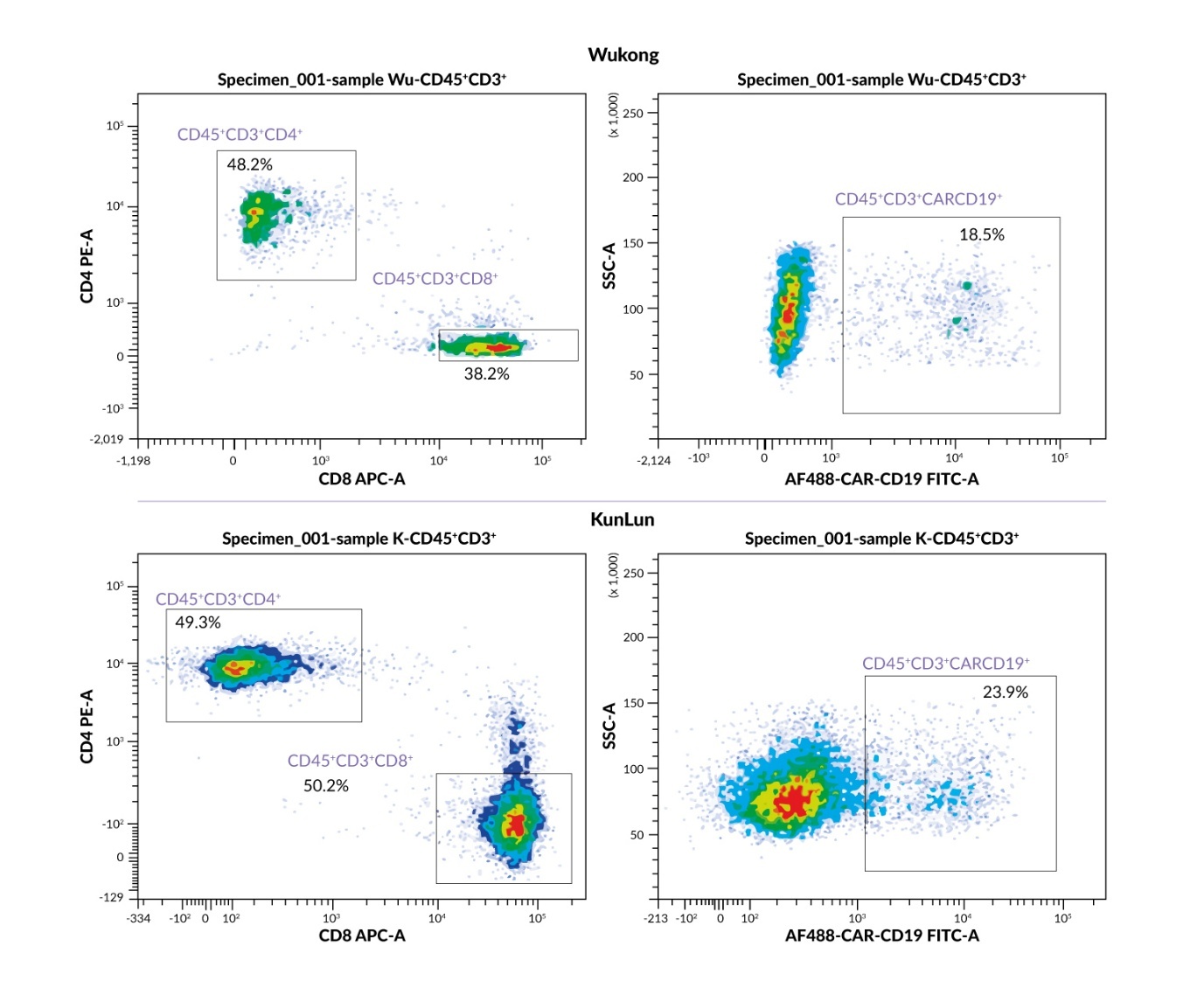
On day 10, the final CD19 CAR T-cell products generated on the Wukong and KunLun platforms were analyzed by flow cytometry to determine product composition and transduction efficiency. We quantified T-cell purity after magnetic enrichment, the proportion of CAR-expressing cells within the CD3+ population and the distribution of CD4+ and CD8+ T-cell subsets in the final products.
After magnetic separation, the CD3+ cell content exceeded 90% for both platforms. Transduction efficiency, defined as the proportion of CD3+ cells expressing the CARCD19 chimeric receptor in the final product, was 18.5% for Wukong and 23.9% for KunLun, indicating slightly higher transduction efficiency of the KunLun platform.
The CD4+/CD8+ subset distribution differed between the platforms. The product obtained on the Wukong platform demonstrated a predominance of T helper (CD3+CD4+) cells over T killer (CD3+CD8+) cells, amounting to 48.2% and 38.3%, respectively. In contrast, the product obtained from the KunLun platform exhibited an almost equal ratio of these subsets, with 49.3% CD4 and 50.2% CD8+ T cells (Figure 4). This difference in the subset balance may have implications for the functional characteristics of the resulting CAR T-cell product.
Evaluation of CAR T lymphocyte functional activity in vitro
To assess CAR T-cell functional activity, effector cells were co-cultured with a CD19+ mKate2-expressing Burkitt’s lymphoma cell line at effector-to-target ratios of 1:1, 5:1 and 10:1. Cytotoxic activity of CAR T cells was analyzed using an Image ExFluorer system (LCI, Korea) after 12 and 24 hours.
The number of viable target cells decreased throughout the entire period of coculture with CAR T lymphocytes. After 12 hours, specific elimination of target cells was observed; however, cytolysis did not exceed 40% for either product even at the highest effector-to-target ratio of 10:1.
By 24 hours, the cytotoxic potential of the CAR T cells was fully realized. The product obtained on the KunLun platform demonstrated higher cytotoxic activity than the Wukong product across all tested ratios, with a maximum specific lysis reaching 78% at a 10:1 ratio. The nonspecific cytotoxicity of unmodified T cells used as a control did not exceed 20% at any time point, confirming the specificity of CAR-mediated cytolysis (Figure 5).
Discussion
This study provides a head-to-head comparison of two closed, automated platforms for CD19 CAR T-cell manufacturing, using the same leukapheresis starting material and an aligned 10-day process. We demonstrated that the automated Wukong and KunLun systems can successfully generate functionally active CD19 CAR T cells that meet key quality criteria, including high viability, high post-selection CD3+ purity, measurable CAR expression and antigen-specific killing of CD19+ target cells, confirming the feasibility of implementing standardized point-of-care manufacturing in clinical practice.
A comparative analysis of the two final products demonstrated differences in manufacturing output despite similar workflow steps, indicating that platform-specific process implementation can influence end-to-end recovery even within closed, automated workflows. The Wukong platform product contained a total cell population of 2.64 × 108 cells, of which 98.3% were CD45+CD3+ T lymphocytes. CAR expression on these cells reached 18.5%, corresponding to 5 × 107 CAR-positive cells. The product produced on the KunLun system was characterized by high CD3+ T-lymphocyte purity of up to 99.4% and higher cell mass accumulation efficiency compared with the Wukong platform, with an approximately two-fold higher total cell yield of 5.11 × 108 cells. CAR expression in the KunLun product was 23.9% of CD3+ lymphocytes, corresponding to 12.2 × 107 CAR+ cells, resulting in a higher absolute number of genetically modified cells at harvest.
The product obtained using the KunLun system was also characterized by a balanced ratio of CD4+ and CD8+ subsets (approximately 1:1), whereas the Wukong platform product showed a predominance of T helper (CD4+) cells, a compositional difference that may be relevant for downstream functional performance. Cell viability consistently exceeded 90% on both platforms.
Functionally, both products mediated specific cytolysis of CD19+ targets, with limited lysis at 12 hours and substantially increased killing by 24 hours, consistent with time-dependent engagement and accumulation of effector mechanisms in co-culture assays. Under the tested conditions, KunLun-derived CAR T cells achieved higher cytolysis rates across effector-to-target ratios, reaching 78% specific lysis at a ratio of 10:1 at 24 hours. The low nonspecific cytotoxicity observed with unmodified T cells supports CAR-dependent activity and argues against assay artifacts associated with coculture stress or imaging readout.
These findings are consistent with previous reports showing that closed, automated platforms can reproducibly generate functional CD19 CAR T-cell products at or near the point of care, including other automated systems such as the CliniMACS Prodigy.2,4 Although this study was not designed to mechanistically dissect the contribution of individual platform characteristics to the final output, the operational differences summarized in Table 1 offer plausible process-level explanations for the observed differences in cell recovery during culture and harvest. In particular, KunLun integrates centrifugation within the culture chamber and therefore avoids transfer of the cell suspension for centrifugation, whereas Wukong requires transfer from the culture chamber to the centrifuge, which can increase the risk of cell loss, particularly late in culture when volumes and cell densities are higher. Such design features are increasingly recognized as practical determinants of end-to-end recovery in closed-system CAR T-cell manufacturing, together with other variables including working volume ranges, sampling strategy and gas control. At the same time, the present comparison underscores that automation is not a uniform intervention: differences in platform architecture (integrated versus separated unit operations), sampling modality and fluidics/centrifugation handling can influence yield, composition and functional readouts even when nominal process steps are similar.
This study has limitations that should be considered when interpreting platform differences. The comparison was performed using starting material from a single donor for the following reasons. First, obtaining a sufficient leukocyte yield requires leukapheresis, an invasive and time-consuming procedure performed under strict clinical control. At our center, leukapheresis material is typically collected for clinical applications only; therefore, additional collections solely for research purposes were not feasible. Second, consumables and reagents required for automated CAR T-cell manufacture, including single-use sets, specialized media, cytokines and lentiviral vectors, are expensive and expanding the study to multiple donor samples would have substantially increased overall costs. Third, using a single donor for both platforms minimized inter-donor biological variability, thereby enabling a more direct assessment of platform- and protocol-associated effects rather than differences attributable to patient-to-patient variations. Future studies using additional donors, replicate runs and deeper product profiling (such as memory/effector phenotypes, exhaustion markers, vector copy number and cytokine secretion) would help distinguish platform effects from run-to-run biological variability and clarify which parameters best predict downstream potency and, ultimately, clinical performance.
Conclusions
In summary, although both the Wukong and KunLun systems supported closed, automated manufacturing of CD19 CAR T cells, the KunLun platform demonstrated advantages across key parameters, including higher cell yield, higher transduction efficiency, a more balanced CD4+:CD8+ subset ratio and higher in vitro cytotoxic activity. These findings support the feasibility of implementing a standardized, automated manufacturing approach in clinical practice and provide a rationale for further preclinical and clinical evaluation. To optimize CAR T-cell therapy with respect to efficacy, safety and accessibility, future studies should define the process factors and platform-specific features that influence in vivo expansion, persistence and clinical efficacy of the resulting CAR T-cell products.
Conflict of interest
The authors declared that the study was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The authors declared that no financial support was received from any organization for the submitted work.
Author contributions
All authors contributed to and approved the final manuscript.
AI use
Artificial intelligence (AI)-assisted tools were used exclusively for the final language editing to improve grammar and readability. These tools were not used to generate scientific ideas or content, analyze data, interpret findings or formulate conclusions. The authors take full responsibility for the accuracy, integrity and originality of the manuscript.
_and_kunlun_(b)_cell_processing_systems.jpeg)
_t-cell_production_using_t.jpeg)
_t_lymphocytes_*in.jpeg)
_and_kunlun_(b)_cell_processing_systems.jpeg)
_t-cell_production_using_t.jpeg)
_t_lymphocytes_*in.jpeg)