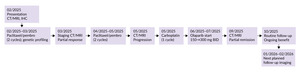
Introduction
Triple-negative breast cancer (TNBC) is an aggressive subtype of breast cancer characterized by the absence of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2) expression.1,2 It is associated with early metastatic spread, limited targeted treatment options and poor prognosis, particularly in the presence of visceral or central nervous system (CNS) involvement. Brain metastases are reported in up to 50% of patients with TNBC, contributing substantially to morbidity and mortality.3 Since TNBC lacks hormone receptor expression and HER2 amplification, endocrine therapy and HER2-directed agents are ineffective and systemic management has historically relied on cytotoxic chemotherapy. Despite its broad use, chemotherapy alone has been associated with modest response rates and limited durability of benefit, with short progression-free intervals and overall survival.4–6
The introduction of immune checkpoint inhibitors has expanded first-line treatment options for patients with metastatic TNBC, with chemoimmunotherapy combinations such as pembrolizumab plus taxane-based chemotherapy becoming the standard of care for tumors expressing programmed death-ligand 1 (PD-L1).7,8
Mutations in tumor suppressor genes BRCA1 and BRCA2 present additional therapeutic challenges in TNBC, as this biologically distinct subset, despite retaining sensitivity to certain chemotherapy agents such as platinum-based regimens, frequently develops resistance to conventional systemic treatments, particularly in the advanced disease setting.1,9,10 Poly (ADP-ribose) polymerase (PARP) inhibitors have emerged as a promising targeted treatment for BRCA-mutated TNBC, exploiting vulnerabilities in the homologous recombination repair pathway.11–13 The PARP inhibitor olaparib has demonstrated clinically meaningful activity in germline BRCA1/2 (gBRCA1/2)-mutated, HER2-negative breast cancer, including triple-negative disease, across different clinical settings.14–18 Furthermore, the clinical efficacy of PARP inhibition in tumors harboring somatic BRCA1/2 (sBRCA1/2) mutations has been established by the phase II TBCRC 048 trial, which demonstrated confirmed responses in patients with metastatic breast cancer treated with olaparib.19,20 These findings extend the therapeutic relevance of PARP inhibitors beyond germline alterations and provide a rationale for their use in sBRCA-mutated TNBC.
Here, we report the case of a patient with disseminated TNBC and extensive intracranial metastases harboring a pathogenic sBRCA2 alteration who was treated with olaparib after progression on first-line chemoimmunotherapy, resulting in sustained clinical benefit.
Clinical Presentation
Initial presentation and staging
A 55-year-old woman presented in February 2025 with severe postprandial lower abdominal pain and a painful, suppurating lesion in the right breast. A right breast mass had been known since May 2024. The clinical course and treatment timeline are summarized in Figure 1.
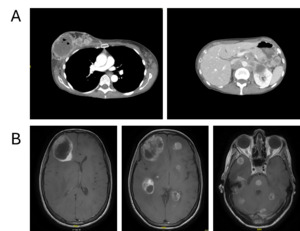
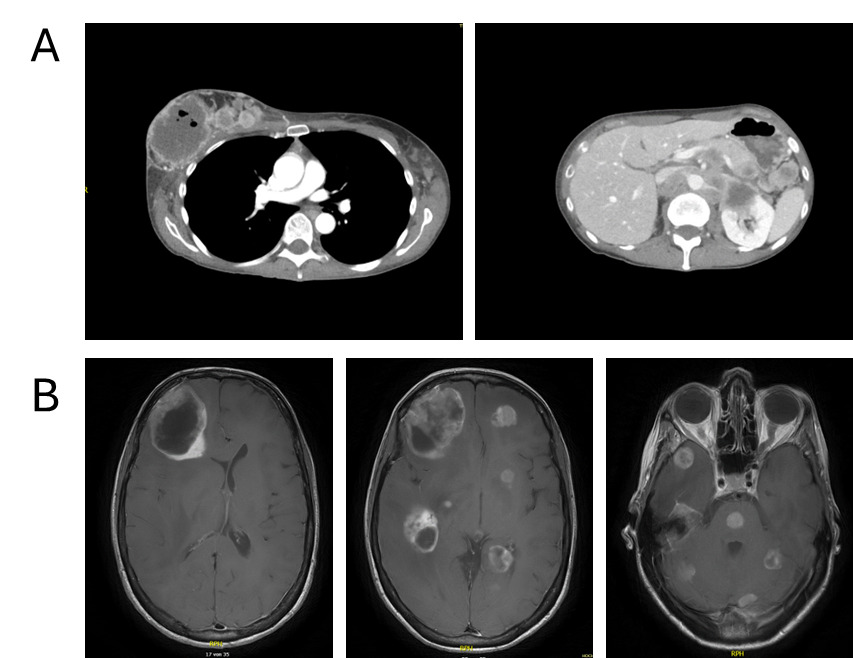
Contrast-enhanced computed tomography (CT) of the thorax and abdomen revealed a right breast mass measuring up to 7 cm, characterized by central necrosis, intralesional gas, peripheral contrast enhancement and cutaneous infiltration (Figure 2A). Extensive lymphadenopathy was present in the bilateral axillary, cervical, supraclavicular and mediastinal regions. Widespread abdominal involvement was noted, including bulky retroperitoneal and pararenal lymphadenopathy with infiltration of the renal hilum and Gerota’s fascia, encasement of major abdominal vessels without occlusion, pancreatic and bilateral adrenal lesions, peritoneal nodules and minimal ascites. No pulmonary, hepatic or suspicious osseous metastases were detected. Overall, the clinical findings were consistent with advanced metastatic breast cancer originating from the right breast and presenting as a disseminated malignancy.
Emergency hospitalization was necessitated due to a rapid deterioration of the patient’s general condition and significant neurological deficits, specifically characterized by somnolence, disorientation and a loss of decisional capacity. Contrast-enhanced CT of the neurocranium revealed multiple (at least 12) supra- and infratentorial intracranial lesions, some partially cystic, associated with extensive perifocal edema, significant mass effect and a leftward midline shift of up to 1.1 cm, with subfalcine and incipient uncal herniation. A metastasis-suspicious left parietal osseous lesion with epidural and subgaleal extension was also identified. Subsequent magnetic resonance imaging (MRI) confirmed extensive cerebral involvement with more than 20 intradural metastases in the frontal and temporal lobes, cerebellum, basal ganglia and pons (Figure 2B). Additional metastatic sites included the left parietal calvarium with extraosseous extension, as well as in the right parotid gland and right masticator space.
Histopathology and molecular diagnostics
Ultrasound-guided core needle biopsies of the right breast mass and left axillary lymph node demonstrated predominantly necrotic invasive breast carcinoma, histologic grade 3 (BRE score 8/9). Immunohistochemistry (IHC) showed the absence of ER and PR expression (0% of tumor cells) and no HER2 overexpression (IHC score 0), consistent with a triple-negative phenotype. The Ki-67 index was 63%.
Axillary lymph node biopsy revealed dense infiltration by poorly differentiated carcinoma with an identical triple-negative immunophenotype, a higher Ki-67 index of approximately 74% and positivity for E-cadherin and cytokeratin 5, supporting basal-like differentiation. IHC analysis of the axillary lymph node metastasis for PD-L1 demonstrated a tumor proportion score (TPS) of 0%, a combined positive score (CPS) ≥20 and an immune cell score IC2.
Diagnosis
Based on the clinical presentation, imaging and histopathological findings, the patient was diagnosed with disseminated poorly differentiated triple-negative breast carcinoma with extensive lymphatic, peritoneal, visceral and CNS metastases.
Therapeutic interventions and outcome
Due to the evidence of more than 10 brain lesions, as well as clinical and neurological worsening of symptoms, the patient initially received whole-brain radiotherapy consisting of five fractions of 4 Gy each in the inpatient hospital setting.
Following a multidisciplinary tumor board discussion, first-line palliative therapy with paclitaxel and pembrolizumab was initiated in February 2025, according to the KEYNOTE-355 regimen.21 A total of four cycles of paclitaxel were administered weekly (120 mg absolute, three doses per cycle), along with four cycles of pembrolizumab (200 mg absolute, once every three weeks).
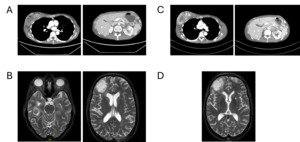
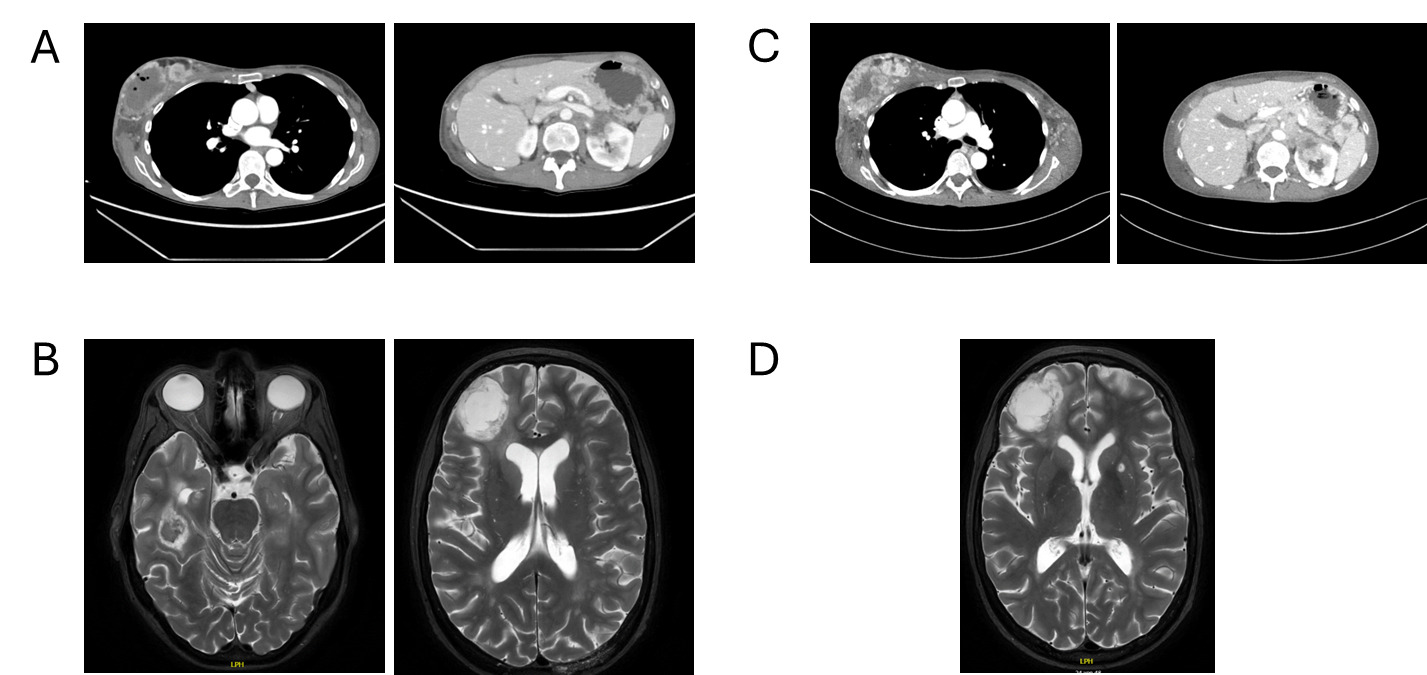
Follow-up in March 2025 showed a marked treatment response after two cycles, with significant regression of intracranial metastases on brain MRI, including resolution of midline shift and cerebral edema, as well as pronounced regression of nodal and visceral disease on contrast-enhanced CT of the thorax and abdomen (Figures 3A and 3B). No new metastatic lesions were identified, consistent with substantial partial remission.
Comprehensive molecular profiling was performed using a circulating tumor (ct)DNA–based next-generation sequencing assay (FoundationOne® Liquid CDx), covering more than 300 cancer-related genes, including seven routinely assessed in this tumor type (AKT1, BRCA1, BRCA2, ERBB2, ESR1, PIK3CA and PTEN). The analysis identified a pathogenic sBRCA2 splice-site mutation (c.8487+1G>A; variant allele frequency 0.81%). Additional genomic alterations included PTEN C105Y, PIK3R1 K561_S565del, FAS splice-site mutation (c.334+1G>T) and TP53 P151S. Microsatellite instability was not detected.
The patient repeatedly declined genetic counselling as a prerequisite for testing of a highly suspected gBRCA2 mutation.
During ongoing treatment, exulceration of the right breast primary tumor developed, indicating an early sign of further disease progression. Initially, it was managed conservatively with oral amoxicillin plus clavulanic acid and referred to specialized wound care. In May 2025, after four cycles of treatment, contrast-enhanced CT and cerebral MRI revealed disease progression involving systemic and intracranial sites (Figures 3C and 3D). Concurrently, thrombosis of the left external iliac vein and new segmental pulmonary emboli in the left lower lobe were detected and therapeutic anticoagulation with apixaban was initiated. Given the radiologic progression on paclitaxel plus pembrolizumab, carboplatin monotherapy was started as an interim measure while reimbursement approval for targeted therapy was pending. However, chemotherapy was discontinued after a single administration at the patient’s request.
Due to the presence of the pathogenic sBRCA2 alteration, targeted therapy with the PARP inhibitor olaparib was initiated in mid-June 2025 at a reduced dose of 150 mg twice daily, which was escalated to the full dose of 300 mg twice daily from July 2025. Treatment was well tolerated without clinically relevant laboratory abnormalities or treatment-limiting adverse events and was continued without dose reduction.
The patient remained clinically stable without new neurological deficits, reporting only mild progressive generalized pain. The ulceration of the right breast primary tumor healed completely with continued wound care and systemic therapy.
Follow-up
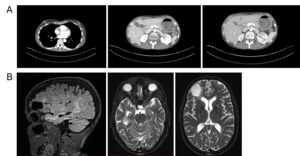
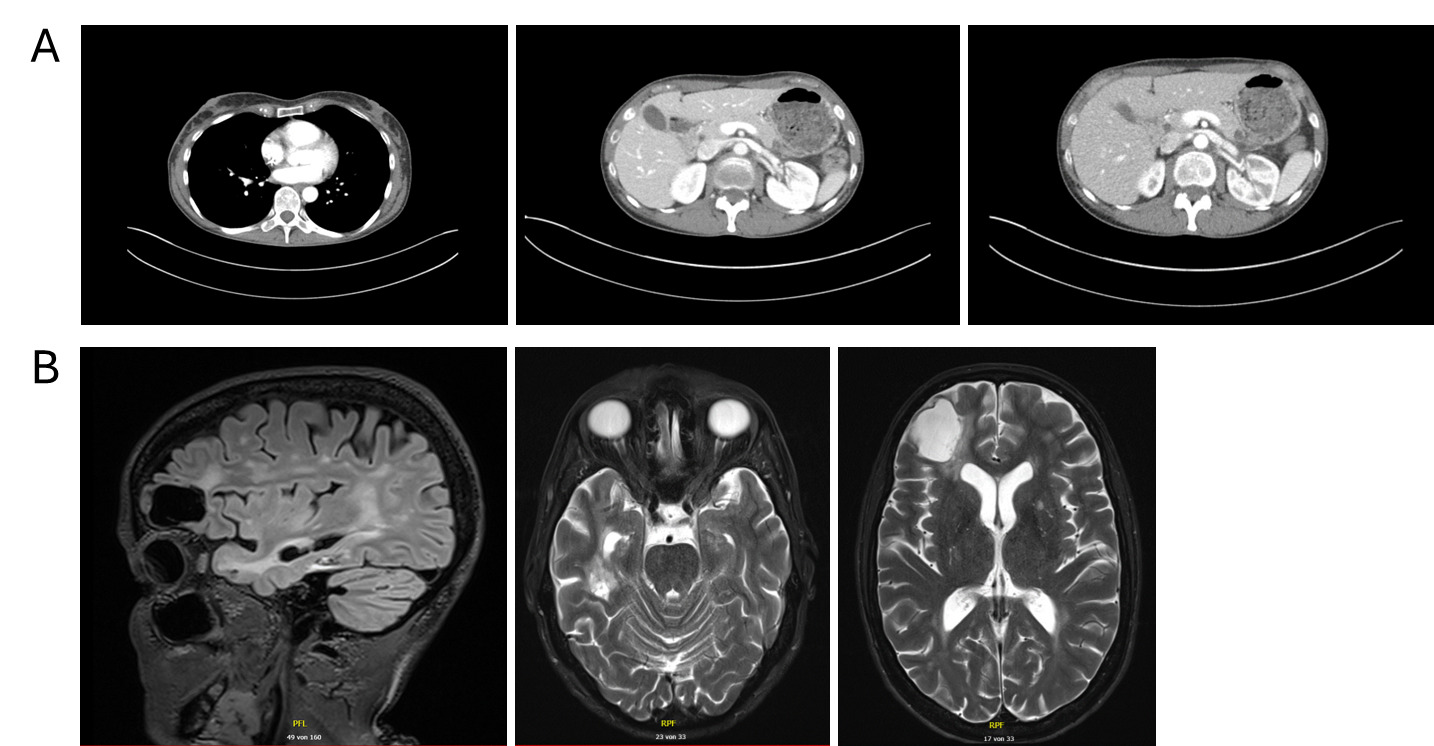
Restaging in September 2025 with contrast-enhanced CT of the thorax and abdomen and contrast-enhanced MRI of the brain demonstrated partial remission, with regression of systemic and intracranial disease and no new metastatic lesions (Figure 4). Therapeutic anticoagulation with apixaban was continued without any new thromboembolic events.
At the last routine follow-up in October 2025, olaparib was continued with ongoing clinical and radiologic benefit. Further imaging is scheduled for early 2026.
Discussion
This case highlights several clinically relevant aspects of managing advanced TNBC, particularly in the context of extensive CNS involvement. The patient demonstrated a pronounced initial response to first-line chemoimmunotherapy according to the KEYNOTE-355 regimen,21 including marked regression of extensive intracranial metastases. This observation supports the activity of pembrolizumab-based combinations in patients with a high tumor burden and CNS involvement following brain radiotherapy, potentially mediated by radiation-induced disruption of the blood–brain barrier, a population that has been underrepresented in pivotal clinical trials.
Despite this early response, disease progression occurred after several cycles of therapy, underscoring the aggressive biology of metastatic TNBC and the limited durability of response frequently observed with first-line chemo-immunotherapy. This progression highlights the ongoing need for additional systemic treatment options in patients who relapse after initial benefit, particularly when durable CNS control is required.
Comprehensive molecular profiling performed during ongoing treatment identified a pathogenic sBRCA2 splice-site mutation, enabling a biologically rational transition to targeted therapy with the PARP inhibitor olaparib. Importantly, olaparib was initiated after radiologic progression on chemoimmunotherapy rather than as maintenance following response, reflecting real-world treatment sequencing rather than trial-defined pathways. Under olaparib therapy, the patient again responded to treatment with regression of both systemic and intracranial disease and sustained clinical benefit with good tolerability. The intracranial response observed under PARP inhibition is clinically noteworthy, as many systemic agents demonstrate limited CNS activity, particularly in patients with a high baseline intracranial tumor burden who have already undergone whole-brain radiotherapy. Therefore, this case adds to the emerging real-world evidence supporting CNS activity and the feasibility of PARP inhibitors in BRCA-mutated TNBC.
Beyond oncologic outcomes, this case also illustrates important aspects of supportive and multidisciplinary care, including the management of cancer-associated thrombosis with apixaban and the successful local treatment of an ulcerated breast lesion, which healed under systemic disease control. Although the role of PARP inhibitors is well established in gBRCA-mutated breast cancer, repeated recommendations for genetic counseling and germline testing were declined by the patient, despite the potential implications for family members.
Conclusions and take-home message
In summary, this case underscores the importance of early and comprehensive molecular testing in metastatic TNBC and demonstrates that flexible, dynamic integration of targeted therapies based on evolving disease characteristics can yield meaningful and durable clinical benefit, even after progression on standard first-line treatment. Prospective studies are needed to better define optimal treatment sequencing and clarify the role of PARP inhibitors in patients previously treated with immune checkpoint inhibitors, particularly with respect to CNS efficacy.
Ethics approval and consent to participate
Written consent for the further use of patient data was obtained.
Consent for publication
Consent for publication was obtained.
Availability of data and materials
All patient data that support this case report are included in anonymized form in the published article.
Conflict of interest
The authors declared that the study was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The preparation of this article was financially supported by AstraZeneca AG. AstraZeneca did not have any decision-making role in the development of the manuscript and did not influence its content in any way.
Authors contributions
All authors contributed to and approved the final manuscript.