CAR T-cell therapy in DLBCL
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma (NHL), accounting for approximately 20–50% of adult NHL cases worldwide.1 It is a biologically and clinically heterogeneous disease, characterized by aggressive clinical behavior, rapid proliferation, and variable genetic and molecular features. While first-line chemoimmunotherapy, such as rituximab combined with cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), achieves a cure in a majority of patients, up to 40% either relapse or are refractory to initial therapy.2,3 Recently, novel bispecific antibodies such as glofitamab, a CD20×CD3 T-cell engaging bispecific antibody, have demonstrated encouraging clinical activity in relapsed or refractory (R/R) DLBCL.4,5 Regimens including glofitamab plus R-CHOP (Glofi-R-CHOP) or the combination of glofitamab with polatuzumab vedotin, a CD79b-targeted antibody-drug conjugate, plus cyclophosphamide, doxorubicin, and prednisone (Glofi-Pola-CHP) are being investigated for their potential to enhance the depth and durability of response beyond standard chemoimmunotherapy.6–9 In recent years, chimeric antigen receptor (CAR) T-cell therapy has emerged as a transformative approach in the treatment of R/R DLBCL.10,11 Clinical trials and real-world evidence have demonstrated durable remissions and survival benefits in a subset of patients treated with CD19-directed CAR T-cell products, including axicabtagene ciloleucel (axi-cel, Yescarta®),12,13 tisagenlecleucel (tisa-cel, Kymriah®),14–17 and lisocabtagene maraleucel (liso-cel, Breyanzi®).18–21 These therapies are now the standard of care for patients who relapse after at least two prior lines of treatment. Based on the landmark trials ZUMA-712,13 and TRANSFORM,19 axi-cel and liso-cel, respectively, have also received approval for use as second-line treatment in R/R DLBCL within 12 months of treatment termination.
Despite their promise, CAR T-cell therapies are associated with many challenges, including manufacturing delays, logistic complexity, and treatment-related toxicity. In particular, the time lag between apheresis and cell infusion, which may span several weeks, poses a risk of disease progression and deterioration of health condition. This highlights the importance of interim disease control strategies, such as bridging therapy, in preserving patient fitness and ensuring successful treatment delivery.
Definition and timing of bridging therapy
Bridging therapy specifically refers to interventions administered after lymphocyte apheresis and before the start of lymphodepleting chemotherapy (Figure 1).22 It is conceptually different from “holding therapy,” which may be used between the decision to refer a patient for CAR T-cell therapy and the collection of lymphocytes. The primary objectives of bridging therapy are not to induce remission but to control disease symptoms, preserve performance status, prevent clinical deterioration, and ultimately keep the patient eligible for lymphodepleting chemotherapy and subsequent CAR T-cell infusion. Patients who do not respond to bridging therapy can still benefit from CAR T-cell therapy and should proceed to infusion as long as they remain fit.
Another key goal of bridging therapy is to lower tumor burden, which is linked to a reduced risk of treatment-related complications such as cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). Therefore, bridging therapy acts both as a temporary measure and as a preparatory intervention to improve the safety profile of subsequent CAR T-cell treatment. Moreover, preserving bone marrow reserve is essential, given the hematologic toxicities (ICAHT) commonly associated with both bridging regimens and subsequent CAR T-cell therapy.
Bridging therapy in pivotal CAR T-cell trials
The design of major phase III trials evaluating CAR T-cell therapies has varied significantly in terms of the allowance for bridging therapy, reflecting different clinical approaches and logistical models (Table 1). In the ZUMA-7 trial, which evaluated axi-cel as second-line therapy for DLBCL, bridging therapy was not permitted, except for corticosteroids.12 This design favored rapid manufacturing and early infusion, minimizing delays between apheresis and treatment. In contrast, the BELINDA trial, which assessed tisa-cel, allowed bridging chemotherapy, and 83% of patients received such therapy.23 Similarly, the TRANSFORM trial, investigating liso-cel and permitting bridging therapy, found that 63% of patients underwent at least one bridging intervention.21
The disparity in outcomes between these trials raises important questions regarding the impact of bridging therapy on efficacy. While ZUMA-7 demonstrated superiority of CAR T-cell therapy over standard of care,12 BELINDA did not meet its primary endpoint.23 A direct trial comparison is not feasible. Nonetheless, it might be tempting to link this difference to the permissiveness of bridging therapy in BELINDA—and, consequently, to the inclusion of patients with more aggressive disease—but such a conclusion would be an oversimplification. Multiple factors, including disease biology, manufacturing times, and baseline patient characteristics confound the relationship between bridging therapy and treatment efficacy.
Real-world use of bridging therapy
Outside the scope of clinical trials, bridging therapy is commonly used. Observational data indicate that 50–90% of patients scheduled for CAR T-cell therapy ultimately receive some form of bridging therapy.24 Different studies have shown inferior outcomes in patients receiving bridging therapy, which probably reflects bridging as a surrogate marker for high-risk disease, rather than a direct negative effect of the therapy itself. A smaller percentage of patients in the United States received bridging therapy compared to those in the United Kingdom (54% vs 87%, respectively).25 This overall high prevalence highlights the challenges of manufacturing delays and the aggressive nature of R/R DLBCL.
However, the prognostic importance of bridging therapy in real-world practice remains controversial. Several retrospective studies have indicated worse outcomes for patients who received bridging therapy.26–29 These results should be interpreted with caution, as bridging therapy is often given to patients with high-risk disease features, such as rapid disease progression, bulky tumors, or poor performance status. In this setting, bridging therapy might serve as a surrogate marker of poor prognosis rather than directly causing the inferior outcomes.
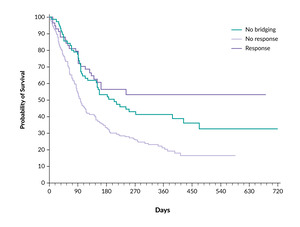
One retrospective US multicenter study evaluated 275 patients undergoing CAR T-cell therapy with axi-cel.30 After applying propensity score matching, the investigators found no statistically significant differences in overall response rate (77% vs 87%; p=0.13), progression-free survival (PFS; HR: 1.25; p=0.23), or overall survival (OS; HR: 1.39; p=0.09) between patients who did and did not receive bridging therapy. Importantly, this study lacked data on response to bridging therapy, which is a critical determinant of subsequent outcomes. Another retrospective German study included over 300 patients, of whom approximately 80% received bridging therapy.31 This analysis demonstrated that patients who responded to bridging therapy had better survival outcomes than those who did not receive bridging therapy or failed to respond. Twelve-month survival rates for patients without bridging, with successful bridging and with bridging failure were 41%, 53% and 20%, respectively (p≤0.001) (Figure 2). These findings suggest that responsiveness to bridging therapy, rather than just its administration, may hold prognostic significance. Similarly, a UK-based retrospective study involving 375 patients showed that a response to bridging therapy, defined as either partial or complete remission, was linked to a 42% reduction in the risk of progression or death after CAR T-cell infusion.32 Notably, polatuzumab vedotin-based combinations seemed especially effective in this group, suggesting that specific treatment plans may provide benefits in the bridging phase.
A recent study investigated the impact of metabolic tumor volume (MTV) response after bridging therapy in 191 patients treated with CAR T cells in the US.33 MTV was determined for all patients on pre-leukapheresis PET and post-bridging therapy/pre-infusion PET. Patients with an improved MTV had a better PFS and less toxicity (i.e. ICANS) compared to patients not responding to bridging therapy. This could help in taking prophylactic measures to reduce toxicity and also serve as prognostic factors. Nevertheless, the question arises as to whether short-term PET CT scans are useful in such situations, and the decision as to whether or not to infuse the cells remains open.
Considerations for individualizing bridging therapy
The decision to initiate bridging therapy and select specific agents must be customized to each patient. Currently, there is no consensus guideline or standard protocol for bridging therapy and practice varies widely based on clinical judgment and institutional experience. Key considerations include the patient’s health status, such as disease symptoms (e.g., local compression and organ involvement), comorbidities, tumor burden, disease progression rate, previous lines of therapy, organ function and manufacturing or vein-to-vein turnaround time.34
In Europe, manufacturing times are generally longer than in the United States, which may require more frequent use of bridging therapy.35 The timing of initiation is crucial; bridging therapy should ideally start immediately after successful apheresis to maximize its effectiveness during the manufacturing period and to ensure adequate disease control while avoiding excessive toxicity that could jeopardize eligibility for lymphodepleting chemotherapy.
Modalities of bridging therapy
A variety of therapeutic options can be used as bridging therapy.25,36 Corticosteroids are often used for symptomatic control and rapid tumor debulking. Radiotherapy plays an important role, particularly in patients with localized disease.37 In some centers, up to 40% of patients receive radiotherapy as part of their bridging approach. Chemoimmunotherapy regimens, such as rituximab-based combinations, remain widely used. Bendamustin should clearly be avoided prior to apheresis (i.e. in the holding setting) but can be used as bridging therapy.38 Polatuzumab vedotin, in combination with bendamustine and rituximab, has shown promising efficacy in the bridging setting, as demonstrated in retrospective cohorts.39,40
As mentioned above, bispecific antibodies, such as CD20xCD3 agents, have recently been approved for R/R DLBCL after two or more prior lines of treatment, demonstrating promising efficacy and tolerability.41 Despite their activity, current evidence and clinical guidelines do not endorse the routine use of bispecific antibodies as bridging therapy before CAR T-cell treatment. Retrospective data suggest that prior exposure to bispecific antibodies does not impair subsequent CAR T-cell efficacy42; however, definitive prospective data on sequencing and optimal integration of bispecific antibodies are currently lacking.
Refining patient selection for bridging therapy
Bridging therapy should be considered for nearly all patients undergoing CAR T-cell therapy, particularly those with high tumor burden, widespread disease, or rapid clinical progression. Observational data and expert consensus indicate that most candidates require some form of intervention during the manufacturing period to prevent clinical deterioration and preserve eligibility for lymphodepleting chemotherapy and CAR T-cell infusion. The decision to initiate bridging therapy should depend on the patient’s condition, disease kinetics and clinical risk factors. It is usually made immediately after successful lymphocyte apheresis to avoid interference with T-cell collection. The choice of bridging regimen must be individualized, aiming to achieve effective disease control while minimizing toxicity. Preservation of immune competence and bone marrow reserve is especially important, as both are critical determinants of CAR T-cell expansion, efficacy and hematologic recovery.
Finally, bridging therapy should be closely coordinated between the referring institution and the CAR T-cell center to ensure alignment in timing, treatment goals, and supportive care planning. This collaborative approach enables timely intervention without delaying downstream CAR T-cell delivery. Although not all patients may need aggressive bridging, it remains an essential part of the treatment pathway for most individuals referred for CAR T-cell therapy.
Future perspectives on bridging therapy
The importance of bridging therapy is likely to change as advances in CAR T-cell technology have shortened manufacturing times and introduced new treatment formats. Rapid manufacturing platforms can reduce or eliminate the need for bridging therapy in some patients. Furthermore, the development of off-the-shelf allogeneic CAR T-cell products43 or in vivo CAR technology44 may render the concept of bridging therapy unnecessary in certain situations.
Nevertheless, even in scenarios with shortened manufacturing timelines, bridging therapy may retain its role as a debulking strategy to optimize subsequent CAR T-cell expansion and function, as well as in reducing CAR T-cell toxicity and complications. The potential synergy between reducing tumor burden and enhancing immune effector engagement requires further investigation.
Conclusions
Bridging therapy remains an integral component of the CAR T-cell treatment pathway for R/R DLBCL. Its primary purpose is to stabilize the disease and keep patients eligible for treatment during the manufacturing period. Although retrospective studies offer conflicting data on its prognostic implications, responsiveness to bridging therapy has consistently emerged as a positive predictor of outcome. There is no standardized approach to bridging therapy; therefore, clinical decisions should be tailored to each patient. Future improvements in manufacturing and new treatment options could change the role of bridging therapy; however, it remains a necessary and evolving tool in managing high-risk lymphoma patients.
Conflict of interest
The authors have declared that the manuscript was written in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The authors have declared that no financial support was received from any organization for the submitted work.
Author contributions
Both authors contributed to and approved the final manuscript.
_t-cell_therapy.png)
_t-cell_therapy.png)